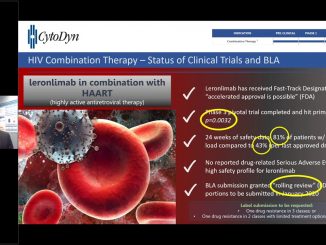
FDA Previously Granted Rolling Review for BLA
VANCOUVER, Washington, Mar 18, 2019 – CytoDyn Inc. (otc.qb:CYDY), (“CytoDyn” or the “Company”) a late stage biotechnology company developing leronlimab (PRO 140), a CCR5 antagonist with the potential for multiple therapeutic indications, today announced that it has filed with the U.S. Food and Drug Administration (FDA) the non-clinical portion of its Biologics License Application (BLA) using the FDA’s Rolling Review process. The non-clinical portion constitutes the first of three sections of the BLA submission for leronlimab (PRO 140) as a combination therapy with HAART for HIV-infected patients. The FDA previously granted Fast Track Designation (FTD) and recently granted Rolling Review for leronlimab (PRO140), which facilitates frequent interactions with the FDA review team. The Rolling Review process allows CytoDyn to submit individual sections of the BLA for review, rather than waiting on FDA review until all three sections are completed and filed.
“This is the most important milestone yet in CytoDyn’s history,” stated Dr. Nader Pourhassan, President, CEO and Director of CytoDyn. “We continue to execute on the submission of our BLA and are well positioned for potential revenue in 2020, subject to final approval. Our team is working diligently to complete the clinical and CMC portions, which represent the remaining two components of the BLA. We are also currently evaluating certain licensing opportunities relating to the commercialization of leronlimab upon approval,” added Dr. Pourhassan.
About Leronlimab (PRO 140)
The U.S. Food and Drug Administration (FDA) has granted a “fast track” designation to leronlimab (PRO 140) as a combination therapy with HAART for HIV-infected patients. Leronlimab (PRO 140) is an investigational humanized IgG4 mAb that blocks CCR5, a cellular receptor that appears to play multiple roles with implications in HIV infection, tumor metastases and immune signaling. Leronlimab (PRO 140) has successfully completed nine Phase 1/2/3 clinical trials in over 700 people, including a successful pivotal Phase 3 trial in combination with standard anti-retroviral therapies in HIV-infected treatment-experienced patients.
In the setting of HIV/AIDS, leronlimab (PRO 140) belongs to a new class of therapeutics called viral-entry inhibitors; it masks CCR5, thus protecting healthy T cells from viral infection by blocking the predominant HIV (R5) subtype from entering those cells. Leronlimab (PRO 140) has been the subject of nine clinical trials, each of which demonstrated that leronlimab can significantly reduce or control HIV viral load in humans. The leronlimab (PRO 140) antibody appears to be a powerful antiviral agent leading to potentially fewer side effects and less frequent dosing requirements compared with daily drug therapies currently in use.
In the setting of cancer, research has shown that CCR5 likely plays a central role in tumor invasion and metastasis and that increased CCR5 expression is an indicator of disease status in several cancers. Moreover, research has shown that drugs that block CCR5 can block tumor metastases in laboratory and animal models of aggressive breast and prostate cancer. CytoDyn is conducting additional research with leronlimab (PRO 140) in the cancer setting and plans to initiate additional Phase 2 human clinical trials, in addition to triple-negative breast cancer, when appropriate.
The CCR5 receptor also appears to play a central role in modulating immune cell trafficking to sites of inflammation and may be crucial for the development of acute graft-versus-host disease (GvHD) and other inflammatory conditions. Clinical studies by others further support the concept that blocking CCR5 using a chemical inhibitor can reduce the clinical impact of acute GvHD without significantly affecting the engraftment of transplanted bone marrow stem cells. CytoDyn is currently conducting a Phase 2 clinical study with leronlimab (PRO 140) to further support the concept that the CCR5 receptor on engrafted cells is critical for the development of acute GvHD and that blocking this receptor from recognizing certain immune signaling molecules is a viable approach to mitigating acute GvHD. The FDA has granted “orphan drug” designation to leronlimab (PRO 140) for the prevention of graft-versus-host disease (GvHD).
About U.S. FDA Fast Track Designation and Rolling Review
The U.S. Food and Drug Administration (FDA)’s Fast Track Designation (FTD) program is designed to expedite clinical development and submission of applications for products with the potential to treat serious or life-threatening conditions and address unmet medical needs. FTD facilitates frequent interactions with the FDA review team, including meetings to discuss the product’s development plan and written communications about issues such as trial design and use of biomarkers. Products that receive FTD, like other products, may be eligible for accelerated approval and priority review if relevant criteria are met. Such products may also be eligible for Rolling Review, which allows a developer to submit individual sections of a product’s applications for review as they are ready, rather than waiting until all sections are complete.
About CytoDyn
CytoDyn is a biotechnology company developing innovative treatments for multiple therapeutic indications based on leronlimab (PRO 140), a novel humanized monoclonal antibody targeting the CCR5 receptor. CCR5 appears to play a key role in the ability of HIV to enter and infect healthy T-cells. The CCR5 receptor also appears to be implicated in tumor metastasis and in immune-mediated illnesses, such as graft-vs-host disease (GvHD) and NASH. CytoDyn has successfully completed a Phase 3 pivotal trial with leronlimab (PRO 140) in combination with standard anti-retroviral therapies in HIV-infected treatment-experienced patients. CytoDyn plans to seek FDA approval for leronlimab (PRO 140) in combination therapy and plans to complete the filing of a Biologics License Application (BLA) in 2019 for that indication. CytoDyn is also conducting a Phase 3 investigative trial with leronlimab (PRO 140) as a once-weekly monotherapy for HIV-infected patients and, plans to initiate a registration-directed study of leronlimab monotherapy indication, which if successful, could support a label extension. Clinical results to date from multiple trials have shown that leronlimab (PRO 140) can significantly reduce viral burden in people infected with HIV with no reported drug-related serious adverse events (SAEs). Moreover, results from a Phase 2b clinical trial demonstrated that leronlimab (PRO 140) monotherapy can prevent viral escape in HIV-infected patients, with some patients on leronlimab monotherapy remaining virally suppressed for more than four years. CytoDyn is also conducting a Phase 2 trial to evaluate leronlimab (PRO 140) for the prevention of GvHD and has received clearance to initiate a clinical trial with leronlimab (PRO 140) in metastatic triple-negative breast cancer. More information is at www.cytodyn.com.
Forward-Looking Statements
This press release contains certain forward-looking statements that involve risks, uncertainties and assumptions that are difficult to predict. Words and expressions reflecting optimism, satisfaction or disappointment with current prospects, as well as words such as “believes,” “hopes,” “intends,” “estimates,” “expects,” “projects,” “plans,” “anticipates” and variations thereof, or the use of future tense, identify forward-looking statements, but their absence does not mean that a statement is not forward-looking. The Company’s forward-looking statements are not guarantees of performance, and actual results could vary materially from those contained in or expressed by such statements due to risks and uncertainties including: (i) the sufficiency of the Company’s cash position, (ii) the Company’s ability to raise additional capital to fund its operations, (iii) the Company’s ability to meet its debt obligations, if any, (iv) the Company’s ability to enter into partnership or licensing arrangements with third parties, (v) the Company’s ability to identify patients to enroll in its clinical trials in a timely fashion, (vi) the Company’s ability to achieve approval of a marketable product, (vii) the design, implementation and conduct of the Company’s clinical trials, (viii) the results of the Company’s clinical trials, including the possibility of unfavorable clinical trial results, (ix) the market for, and marketability of, any product that is approved, (x) the existence or development of vaccines, drugs, or other treatments that are viewed by medical professionals or patients as superior to the Company’s products, (xi) regulatory initiatives, compliance with governmental regulations and the regulatory approval process, (xii) general economic and business conditions, (xiii) changes in foreign, political, and social conditions, and (xiv) various other matters, many of which are beyond the Company’s control. The Company urges investors to consider specifically the various risk factors identified in its most recent Form 10-K, and any risk factors or cautionary statements included in any subsequent Form 10-Q or Form 8-K, filed with the Securities and Exchange Commission. Except as required by law, the Company does not undertake any responsibility to update any forward-looking statements to take into account events or circumstances that occur after the date of this press release.